1253
Views & Citations253
Likes & Shares
Seminal
Plasma (SP) proteins are rich with many proteins of different genital tract
origin so the fields of proteomics were promise for the development of novel
male infertility biomarkers. Seminal plasma proteins Testis Expressed Protein
101 (TEX101) and Extracellular Matrix Protein 1 (ECM1) assay are already
available or under final development for clinical use, so the aim of study,
evaluation of TEX101 and ECM1 Seminal Plasma (SP) proteins for assessment the
predictive of Sperm Retrieval Rate (SRR) in testicular sperm retrieval and
diagnosis obstructive from non-obstructive azoospermia.
A case control study was included 65 infertile
azoospermic men were subjected to clinical examination, seminal fluid analysis,
hormonal investigation and SP proteins TEX101 and ECM1 assessment by Enzyme
Linked Immuno-Sorbent Assay (ELISA) as well as they were subjected to the
conventional Testicular Sperm Extraction (TESE) technique, mincing with searching
for sperm.
The results of study included mean age of 65 men
were recorded 33.37 ± 6.99 years which were divided into 10 (15.38%)
Obstructive Type (OA) and 55 (84.62) Non-Obstructive (NOA) type. The SRR
account 36 out of 65 patients (55.4%) were divided into OA (100%) and NOA
(47.3%) and the difference was significant (P=0.014).
The TEX101 and ECM1 were a significantly (P<0.001
and P=0.007, respectively), higher in
NOA than OA. The receiver operating characteristic curve or ROC curve show that
the SP TEX101 cut-off values above 0.9 ng/ml is candidate to sperm retrieval
technique. The ECM1 protein, the cut-off values (>943.11 pg/ml for
differentiation of NOA versus OA.
Keywords: Proteomics, TEX101, ECM1, Sperm
retrieval technique, Reproductive hormones, Testicular histopathology
INTRODUCTION
Infertility is a common condition among men and women and it is
inability to achieve pregnancy within 12 months of a regular unprotected
intercourse. It is occurred in 15% of the cases of the reproductive aged
couples [1,2]. Infertility may be related to a female factor, a male factor, a
combination of both or it may be unexplained. Two thirds of the cases are
attributed to male factors [3].
The clinical categories of male infertility range from lowered
production of sperm (oligozoospermia) to severe cases of azoospermia with
non-measurable levels of sperm in semen [4].
Azoospermia affects about 1% of all men and 15% of infertile men [5].
It is absent of spermatozoa in the semen sample following the standard seminal
fluid analysis as recommended by the World Health Organization (WHO). When
spermatozoa are absent in the wet preparation, an examination of the
centrifuged sample (3000x g) for 15 min is recommended. Otherwise, no sperm are
observed in the
Azoospermia is
classified to Non-Obstructive (NOA) and Obstructive Azoospermia (OA). Based on
histological evaluation of testicular tissue, the NOA subtype is further
classified into Hypo Spermatogenesis (HS), Maturation Arrest (MA) and Sertoli
Cell-Only syndrome (SCO) [7].
Obstructive azoospermia results from physical
obstruction in the male reproductive tract due to congenital or acquired defects
in the epididymis or vas deferens [8].
The level of
spermatogenesis and the presence of sperm in the testis are diagnosed by
testicular biopsy till now it was a standard tool for differential diagnosis of
azoospermia [9]. However, it is an invasive surgical procedure with potential
complications. So that there is an important need for substitute, non-invasive
procedures for differential diagnosis of azoospermia of male infertility and
further classification of its subtypes.
The SP is derived
from male reproductive organs which was rich with epididymis and testis-derived
proteins, mRNA and metabolites. It has been used as a suitable clinical sample
for the non-invasive diagnosis of a wide range of male reproductive system
disorders [7,10].
The SP composed of
3200 proteins secreted by different genital organ origin like testes, epididymis,
prostate, seminal vesicles and Cowper’s glands and these are directly involved
in the production and maturation of sperm or in the interaction with the zona
pellucida and fusion with oocytes [11-13].
Testis-specific
biomarkers are not found in other biological fluid like blood due to stringent
blood-testis and blood-epididymis barriers, semen and SP remain the only
available fluids for the non-invasive diagnosis of male infertility [14,15].
The new research in
the subject of proteomics may be promised for the advancement of novel male
infertility biomarkers.
The SP protein-based
assessment of Tests Express protein 1 (TEX101) and epididydmal specific protein
1 or Extracellular Matrix protein 1 (ECM1) are already discovered and under
final development for clinical use. Immunoassays of ECM1 and TEX101 have the
potential to roll out most of the histopathological diagnosis of testicular
biopsies and TESE procedures for patients with azoospermia also to facilitate
prediction of the outcome of sperm retrieval procedures used for assisted
reproduction and to reduce the total cost of azoospermia diagnosis.
MATERIALS AND METHODS
A case control study
included 65 infertile azoospermic males were manifested as male factor
infertility (normal female partners) according to history, examination and
investigation, in the period from November 2017 till January 2019 at the Male
Infertility Clinic of High Institute for Infertility Diagnosis and Assisted
Reproductive Technologies, the patients were undergone a detailed history,
clinical examination and laboratory investigation such as seminal fluid
analysis, hormonal and seminal plasma proteins (TEX101, ECM1) assessment and
then they were subjected to the testicular biopsy after written consent of
patients.
Seminal fluid
analysis is performed before and after centrifugation to confirmed azoospermia
as well as seminal plasma collection and freezing to be thawed litter for
assessment seminal plasma proteins TEX101 and ECM1 by Enzyme Linked
Immuno-Sorbent Assay (ELISA).
Testicular biopsies
were planned to be separated into two samples one subjected to mincing and
searching for sperm then cryopreservation to be used for ICSI, the other sample
sent to pathologist for histopathological diagnosis of azoospermic types.
STATISTICAL ANALYSIS
Data were collected,
summarized, analyzed and presented using Statistical Package for Social
Sciences (SPSS) version 23 and Microsoft Office Excel 2010. Qualitative (categorical)
variables were expressed as number and percentage, whereas, quantitative
(numeric) variables were first evaluated for normality distribution using
Kolmogorov-Smirnov test and then accordingly normally distributed numeric
variables were expressed as mean (an index of central tendency) and standard
deviation (an index of dispersion), while those numeric variables that are not
normally distributed were expressed as median (an index of central tendency)
and inter-quartile range (an index of dispersion). The following statistical
tests were used: Chi-square test was use to evaluate association between any
two categorical variables provided that less than 20% of cells have expected
count of less than 5. However, Fischer exact test was used instead when
chi-square test was not valid (in case that more than 20% of cells have
expected count of less than 5). Independent samples t-test was used to evaluate
the difference in mean of numeric variables between any two groups provided
that these variables were normally distributed; otherwise Mann Whitney U test
would be used instead if those variables were not normally distributed. One way
Analysis of Variance (ANOVA) was used to evaluate difference in mean of numeric
variables among more than two groups provided that these numeric variables were
normally distributed; but Kruskal Wallis test was chosen in case of
non-normally distributed variables. One way ANOVA was followed by post-hoc LSD
test to evaluate individual differences in mean values between any two groups
among groups tested primarily using one way ANOVA; whereas, Kruskal Wallis test
was followed by Mann Whitney U test for the same purpose in case of
non-normally distributed numeric variables.
RESULTS
The results of study
included mean age of
65 men were recorded 33.37 ± 6.99 years which were
divided into 10 (15.38%) obstructive type (OA) and 55 (84.62) non-obstructive
(NOA) type as in Table 1.
The SRR account 36 out of 65 patients (55.4%)
were divided into OA (100%) and NOA (47.3%) and the difference was significant
(P=0.014) as in Table 2.
The TEX101 and ECM1 were a significantly (P<0.001
and P=0.007, respectively), higher in NOA than OA as in Table 3. The SP level of TEX101 was statistically highly significant (P=0.005),
being higher in men with positive sperm retrieval 1.48 (1.55) ng/ml than
negative sperm retrieval 0.31 (1.35) ng/ml as shown in Table 4. The ROC
show that the SP TEX101 cut-off values above 0.9 ng/ml is candidate to sperm
retrieval technique with CI equal to 57.8%-81%, Sensitivity (66.7%) and
Specificity (69.0%) as in Table 5 and Figure 1.
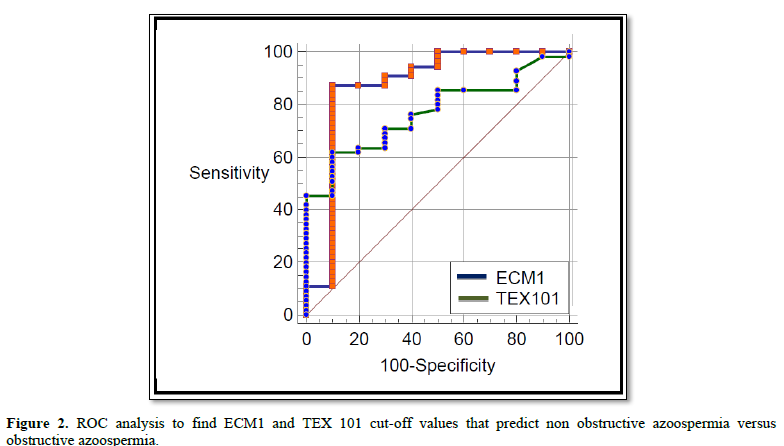
To test the validity
of ECM1 and TEX101 in the differentiation between obstructive and
non-obstructive azoospermia an ROC analysis was carried out and the results are
shown in Figure 2 and Table 6. ECM1
cut-off value was >943.11 pg/ml with a sensitivity rate of 87.3% and
specificity rate of 90%. In addition, the accuracy rate was 87.1%. On the other
hand, TEX101 cut-off value was >0.79 ng/ml with a sensitivity rate of 61.8%
and specificity rate of 90%. Moreover the accuracy rate was 76.9%. In both situations
the level of significance was high (p<0.001).
DISCUSSION
The age of the
patients enrolling in the study was ranging from 22 to 48 years with a mean age
of 33.37 ± 6.99 years which is nearly similar to that reported by other
literature's [16,17] who conducted a study on 60 and 76 azoospermic patients,
they reported a mean age of patients are 33.32 ± 7.55 years and 35.1 ± 60
years, respectively.
In addition to the
other literatures, it was approximately as same the mean age of azoospermic
patients, 35.5 ± 8.30 years and 33.38 ± 7.44 years [18,19] they were studied
451 and 20 azoospermic patients, respectively.
About the type of
azoospermia the presented study was included 15.38% obstructive type and 84.62%
non-obstructive type, which is nearly the same the result of previous literatures
[20-22] the OA is less common than NOA and occurs in 15-20% of men with
azoospermia, whereas other study reported that NOA is diagnosed in 49% to 93%
and post-testicular obstruction or retrograde ejaculation are estimated to
affect from 7% to 51% of azoospermic men of azoospermic patients [23,24].
With respect to OA
(normaspermatogensis) the results of Rashed et al. [25] were approximately
similar to current observation, in which, the cases of normal spermatogenesis
were 15% and 24%, however other study there was a higher incidence (28%) of
normal spermatogenesis [26].
Regardless of the underlying etiology,
management of patients with azoospermia usually relies upon the recovery of
spermatozoa with a testicular biopsy/sperm extraction procedure and a successful
in vitro fertilization with intra-cytoplasmic sperm injection, so that one of the effective
parameters that should be considered in the management of azoospermic patients
is the ability to predict the rate of spermatozoa recovery in these patients.
Understanding these parameters is also important for counseling the patient and
his wife [27].
Regarding the SRRs,
the other study was reported SRRs of 16.7-45% by conventional TESE (cTESE)
[27].
Salehi et al. [28]
showed that the overall mean rate of SRR was 48.8%, which was approximately
same as current observation.
Abdel Rahem et al.
[29] were studied, 112 patients had obstructive azoospermia and 276 patients
had NOA, it reported all patients in the obstructed group had a positive sperm
while the sperm retrieval rate for the NOA group was 50%.which is nearly same
as observation of under current study. The study of Cissen et al. [30] included
599 (43.7%) with successful sperm retrievals after a first TESE procedure of
NOA.
Obstructive
azoospermia is less common than non-obstructive azoospermia and occurs in 15 to
20% of men with azoospermia [31].
Although NOA
indicates impaired sperm production of the entire testis by definition, it has
been observed that focal normal spermatogenesis can be observed in 50 to 60% of
men with NOA [32].
The laboratory
technique, embryologists experience, pathologist, single or multiple, site,
unilateral or bilateral testicular biopsy and type of SR technique are possible
causes of difference in the SRR.
During the last
decades, seminal plasma protein has gained an important role in male
infertility assay and proteomics has been serving as a tool for biological
research of spermatogenesis and the clinical research of male infertility.
One of research biological tool for SP
proteins assay based on the previous measurement by ELISA [33]. The current
study represents the first study of proteomics in IRAQ.
The ELISA technology is used for quantitative
detection of TEX101 which is range 0.313-20 ng/ml and sensitivity <0.188 ng/ml,
whereas ECM1 is range 31-2000 and P=0.007, respectively), higher in men
with non-obstructive azoospermia than men with obstructive azoospermia, which
is similar to other studies that reported, a proteomic analysis of seminal
plasma has shown the absence of certain proteins responsible for sperm function
and proteins were absent in azoospermic patients such as both Seminal plasma
level of ECM1 and TEX101 were significantly higher in men with NOA than men
with OA [34,35].
Drabovich et al. [36] have identified ECM1
and TEX101proteins in seminal plasma that could be help facilitate the
differential diagnosis of azoospermia. Testing such SP, may be able to
distinguish patients with OA and NOA [36].
Proteomic analysis of seminal plasma has
shown the absence of certain proteins in the seminal plasma, however many
proteomic analysis were perform to determine the differential expression of
proteins in azoospermia [37,38].
The result of
presented study is similar to Drabovich et al. [36] was reported that testis-expressed
protein 101 is characterized as the biomarker for azoospermia and extracellular
matrix protein 1 was able to differentiate NOA and post-vasectomy men with a
threshold value of 2.3 ng/mL.
In humans, several
seminal plasma proteins were found which serve as diagnostic markers of
spermatogenesis, seminiferous epithelium state and azoospermia [39].
So that from these previous and current
observation, high SP level of two protein in NOA versus low level in cases of
OA, this fact due to a focal spermatogenesis of deferent score in between NOA
as mention above [33].
According to OA and NOA subtype in the
presented observation, Seminal plasma level of ECM1 was significantly lowest in
men with OA (P<0.05), on the other hand, Seminal plasma level of
TEX101 was significantly lowest in men with OA (P<0.05) so both
proteins were characterized as biomarker for diagnosis OA from NOA. A positive
significant correlation of Seminal plasma level of ECM1 to serum level of FSH,
these result on the same line of other literatures which reported an emerging
SP proteins assay as biomarkers for the noninvasive diagnosis of male
infertility and differentiation of azoospermia forms, OA versus NOA and
histopathological subtypes of the NOA azoospermia [40,41].
Sperm retrieval rat in the current study show
no statistical significant between positive SR versus negative SR with respect
to ECM1, whereas SP level of TEX101 was statistically highly significant (P=0.005),
being higher in men with positive sperm retrieval 1.48 (1.55) ng/ml than
negative SR 0.31 (1.35) ng/ml. However one of researcher reported TEX101 could
differentiate between hypospermatogenesis and sertoli cell-only syndrome (but
not between MA and SCO) with prediction of spermatozoa success rates for the
corresponding subtypes were HS (100%), MA (55%) and SCOS (0%) [33].
Identification of
both testis-specific and germ cell type-specific proteins secreted into semen
exclusively by spermatocytes, spermatids or spermatozoa should provide markers
to accurately pinpoint the stage of spermatogenesis failure and thus predict
TESE outcome with a better diagnostic performance [42].
The levels of ECM1
protein were high in NS (~40 µg/ml) and NOA (~20 µg/ml) samples, but notably
decreased in OA/PV samples (~1 mg/ml).
Post-vasectomy seminal
plasma samples are void of proteins originating from the testis and the
epididymis due to ligation of the vas deferens [43].
When azoospermia is
diagnosed by semen analysis, low SP levels of ECM1 and TEX101 proteins suggest
obstructive azoospermia, while high SP level of ECM1 suggests non-obstructive
azoospermia.
These observations
confirmed that two proteins can be used as diagnostic of choice to
differentiate between OA and NOA [44].
TEX101 is a membrane
protein with specific expression in germ cells only, it is GPI-anchored, mouse
TEX101 is expressed in testis but released from the surface of spermatozoa by
highly specific enzymatic mechanisms during sperm maturation in the epididymis
[45].
These report explain
why the physical obstruction to seminal out flow and the absence of germ cells
lead to very low (theoretically zero) levels of TEX101 in SP of patients with
OA, PV and SCO whereas in in other subtypes of NOA, TEX101 is expressed, but
the male gamete that failed to mature (sperm cells) never pass through the
epididymis to allow for the cleavage of TEX101 from the surface of spermatozoa.
This fact suggested that TEX101 can be released from the spermatocytes membrane
inside the testis by non-specific mechanisms, TEX101 expression per germ cell
may vary in different individuals and TEX101 was released into SP not only by
epididymal spermatozoa, but also by testicular germ cells. So it is detected in
SP in low concentration (<120 ng/ml) this lead to fact, SP concentration of
TEX101 alone allows for the differentiation of histopathological NOA subtypes
which is more specific for differentiated sertoli cell-only syndrome from the
other categories of NOA [46].
These results give an explanation of current
study which reported a high SP TEX101and ECM1 level incases NOA than OA which
are 1.44 (1.63) ng/ml and 1629.10 pg/ml versus 0.22 ng/ml and 469.60 pg/ml,
respectively.
Furthermore, ECM1
levels was higher in fertile men and in men with non-obstructive azoospermia,
but nearly absent in vasectomized men, differentiating these conditions with
high specificity and sensitivity [43], on the other side, TEX101 levels were
higher in fertile men and undetectable in SCOS and post-vasectomy samples [43],
which was similar to current observation. These data may be strengthening the
confidence in non-obstructive azoospermia and obstructive azoospermia diagnosis
using these two SP and gives predictive value of Testicular Sperm Extraction
(TESE) outcome [47].
Receiver Operator
Characteristic (ROC) curve to identify serum TEX101 cut-off values, Area Under
Curve (AUC), Accuracy, 95% Confidence Interval (CI), Sensitivity and
Specificity that predict positive sperm retrieval, so that any patient with
seminal plasma TEX101concentration above 0.9 ng/ml is candidate to sperm retrieval
technique .The prediction of sperm retrieval by TEX101 was comparable to other
study which revealed TEX101 AUC=0.69 (95% CI 0.48-0.89). With the cut-off of ≥
0.6 ng/mL, TEX101 had 73% sensitivity, 64% specificity,70% positive and 68%
negative predictive values [46].
Regarding ECM1, the
ROC curve, the presented observation were nearly same as finding of other
observation were reported that sensitivity, specificity and threshold value
were equal to 100, 73 and >2.3 µg/ml [48]. Whereas, other study reported
that AUC (0.99) with sensitivity equal to 94% and the ECM1 (<2.3 µg/ml)
suggest an OA, but high seminal plasma level of ECM1 (>2.3 µg/ml) suggest
NOA [36] which is approximately same the sensitivity in the current study.
CONCLUSION
Although late; but
the first an Iraqi study from which it can conclude and focus light on the
followings:
It should be noted
that seminal plasma TEX101 and ECM1 proteins are promising to be differentiated
between OA/NOA and predict the success of sperm retrieval especially when
complemented with testing reproductive hormones like a follicular stimulating
and luteinizing hormone while TEX101 SP alone was moderate predictive value for
diagnosis of NOA subtypes and SRR but unconventional alone for clinical
diagnostics. From presented observation that including testicular
histopathology patterns, method of TESE surgery and seminal plasma proteins,
may be able to predict the chances of obtaining spermatozoa in patients with
azoospermia. Although, in despite of the efficiency of some predictive
procedures, no one of them are superior to other.
AUTHORS CONTRIBUTION STATEMENT
This research was
done by MB Ch. B, DGS, M. ART's student Huaasin Khaleefa Kadhem Al Dulaimy as a
part of his thesis under the supervision of Prof. Dr. Ula Al Kawaz and Assist.
Prof. Dr. Hayder AL Mossa (corresponding author).
ACKNOWLEDGEMENT
The researchers are
thankful for the co-worker.
CONFLICT OF INTEREST
Conflict of interest
declared none.
1.
(2011) Male Infertility Best Practice Policy
Committee of the American Urological Association, Practice Committee of the
American Society for Reproductive Medicine Report on evaluation of the
azoospermic male. AUA. Education and Research, Inc.®, pp: 1-20.
2.
Stephens SM, Arnett DM, Meacham RB (2013) The use
of in vitro fertilization in the management of male infertility: What the
urologist needs to know? Rev Urol 15: 154-160.
3.
Slama R, Hansen O, Ducot B, Sorensen D, Giorgis
Allemand L, et al. (2012) Estimation of the frequency of involuntary
infertility on a nation-wide basis. Hum Reprod 27: 1489-1498.
4.
Mosher WD, Pratt WF (1991) Fecundity and
infertility in the United States: Incidence and trends. Fertil Steril 56:
192-193.
5.
Muhammad H, Shah AA, Nabi G (2015) Male
infertility: Etiological factors (a review). Am Eurasian J Toxicol Sci 7:
95-103.
6.
World Health Organization (2010) WHO laboratory
manual for the examination and processing of human semen. 5th Edn.
Geneva: World Health Organization, p: 47.
7.
McLachlan RI, Rajpert-De Meyts E, Hoei-Hansen CE,
de Kretser DM, Skakkebaek NE (2007) Histological evaluation of the human testis
- Approaches to optimizing the clinical value of the assessment: Mini review.
Hum Reprod 22: 2-16.
8.
Cocuzza M, Alvarenga C, Pagani R (2013) The
epidemiology and etiology of azoospermia. Clinics 68: 15-26.
9.
Dohle GR, Elzanaty S, van Casteren NJ (2012)
Testicular biopsy: Clinical practice and interpretation. Asian J Androl 14:
88-93.
10.
Carpi A, Sabanegh E, Mechanick J (2009)
Controversies in the management of non-obstructive azoospermia. Fertil Steril
91: 963-970.
11.
Muttukrishna S, Yussoff H, Naidu M, Barua J,
Arambage K, et al. (2007) Serum anti-Mullerian hormone and inhibin B in
disorders of spermatogenesis. Fertil Steril 88: 516-518.
12.
Brewis IA, Gadella BM (2010) Sperm surface
proteomics: From protein lists to biological function. Mol Human Reprod 16:
68-79.
13.
Heshmat SM, Mullen JB, Jarvi KA, Soosaipillai A,
Diamandis EP, et al (2008) Seminal plasma lipocalin-type prostaglandin D
synthase: A potential new marker for the diagnosis of obstructive azoospermia.
J Urol 179: 1077-1080.
14.
Heriberto, Rodrı´guez-Martı´nez, UlrikKvist, Jan
Ernerudh, LibiaSanz, et al. (2011) Seminal plasma proteins: What role do they
play? Am J Reprod Immunol 66: 11-22.
15.
De Hoog CL, Mann M (2004) Proteomics. Annu Rev
Genomics Hum Genet 5: 267-293.
16.
Al Kawaz Ula, Sally A, Ban JQ, Mohammad OS (2016)
Immunohistochemical expression of MCL-1 in testicular biopsy of patient with
azoospermia. Int J Adv Res 4: 1759-1767.
17.
Jorsaraei SGA, Shafi H, Alereza H (2016)
Azoospermia and testicular biopsy before intracytoplasmic sperm injection: Does
the type of anesthesia make a difference? J Nat Sci Biol Med 7: 89-92.
18.
Eisenberg ML, Shy M, Walters RC (2012) The
relationship between anogenital distance and azoospermia in adult men. Int J
Androl 35: 726-730.
19.
Ozkavukcu S, Ibis E, Kizil S, Isbacar S, Aydos K
(2014) A laboratory modification to testicular sperm preparation technique
improves spermatogenic cell yield. Asian J Androl 16: 852-857.
20.
Rizk BR, Aziz N, Agarwal A, Edmund SJ (2014)
Medical and surgical management of male Infertility. 1st Edn. New
Delhi: Jaypee 15: 117-119.
21.
Jungwirth A, Diemer T, Dohle GR, Giwercman A, Kopa
Z, et al. (2012) European Association of Urology Guidelines on Male
Infertility. Eur Urol 62: 324-332.
22.
Schlegel PN (2004) Causes of azoospermia and their
management. Reprod Fertil Dev 16: 561-572.
23.
Pastore A, Palleschi G (2012). Male infertility.
InTech Obstructive and Non-Obstructive Azoospermia 1: 1-20.
24.
Matthew W, Marc G, Hardy MP (2014) Review of
azoospermia. Landes Bioscience Spermatogenesis 4: e28218-7.
25.
Rashed MM, Ragab NM, Shalaby AR, Ragab WK (2008)
Patterns of testicular histopathology in men with primary infertility. Internet
J Urol 2: 1-4.
26.
Practice Committee of American Society for
Reproductive Medicine in collaboration with Society for Male Reproduction and
Urology (2018) The management of infertility due to obstructive azoospermia.
Fertil Steril 109: 777-782.
27.
Deruyver Y, Vanderschueren D, Van der Aa F (2014)
Outcome of microdissection TESE compared with conventional TESE in
non-obstructive azoospermia: A systematic review. Andrology 2: 20-24.
28.
Salehi P, Derakhshan-Horeh M, Nadeali Z,
Hosseinzadeh M, Sadeghi E, et al. (2017) Factors influencing sperm retrieval
following testicular sperm extraction in non-obstructive azoospermia patients.
Clin Exp Reprod Med 44: 22-27.
29.
Abdel Raheem A, Garaffa G, Rushwan N, De Luca F,
Zacharakis E, et al. (2012) Testicular histopathology as a predictor of a positive
sperm retrieval in men with non-obstructive azoospermia. BJU 111: 492-499.
30.
Cissen M, Meijerink AM, D’ Hauwers KW, Meissner A,
van der Weide N, et al. (2016) Prediction model for obtaining spermatozoa with
testicular sperm extraction in men with non-obstructive azoospermia. Hum Reprod
31: 1934-1941.
31.
Jungwirth A, Diemer T, Dohle GR, Giwercman A, Kopa
Z, et al. (2012) European Association of Urology Guidelines on Male
Infertility. Eur Urol 62: 324-332.
32.
Gnessi L, Scarselli F, Minasi MG, Mariani S,
Lubrano C, et al. (2018) Testicular histopathology, semen analysis and FSH,
predictive value of sperm retrieval: supportive counseling in case of
reoperation after testicular sperm extraction. BMC Urol 18: 63.
33.
Dimitrios K, Schiza C, Brinc D, Soosaipillai A,
Theano DK (2017) Preclinical evaluation of a TEX101 protein ELISA test for the
differential diagnosis of male infertility. BMC Med 15: 60.
34.
Selvam MKP, Agarwal A (2018) Update on the
proteomics of male infertility: A systematic review. Arab J Urol 16: 103-112.
35.
Zhou T, Zhou ZM, Guo XJ (2013) Bioinformatics for
spermatogenesis: Annotation of male reproduction based on proteomics. Asian J
Androl 15: 594-602.
36.
Drabovich AP, Dimitromanolakis A, Saraon P,
Soosaipillai A, Batruch I, et al. (2013) Differential diagnosis of azoospermia
with proteomic biomarkers ECM1 and TEX101 quantified in seminal plasma. Sci
Transl Med 5: 212 ra160.
37.
Yamakawa K, Yoshida K, Nishikawa H, Kato T, Iwamoto
T (2007) Comparative analysis of inter-individual variations in the seminal
plasma proteome of fertile men with identification of potential markers for
azoospermia in infertile patients. J Androl 28: 858-865.
38.
Drabovich AP, Jarvi K, Diamandis EP (2011)
Verification of male infertility biomarkers in seminal plasma by multiplex
selected reaction monitoring assay. Mol Cell Proteomics 10: 1-13.
39.
Mogielnicka-Brzozowska M, Kordan W (2011)
Characteristics of selected seminal plasma proteins and their application in
the improvement of the reproductive processes in mammals. Polish J Vet Sci 14:
489-499.
40.
Drabovich AP, Martinez-Morillo E, Diamandis EP
(2015) Toward an integrated pipeline for protein biomarker development. Biochim
Biophys Acta 1854: 677-686.
41.
Drabovich AP, Pavlou MP, Batruch I, Diamandis EP
(2013) Proteomic and mass spectrometry technologies for protein biomarker
discovery. In: Isaaq HJ, Veenstra TD, editors. Proteomic and Metabolomic
Approaches to Biomarker Discovery. Amsterdam: Elsevier, p: 472.
42.
Bieniek JM, Drabovich AP, Lo KC (2016) Seminal
biomarkers for the evaluation of male infertility. Asian J Androl 18: 426-433.
43.
Batruch I, Lecker I, Kagedan D, Smith CR, Mullen
BJ, et al. (2011) Proteomic analysis of seminal plasma from normal volunteers
and post-vasectomy patients identifies over 2000 proteins and candidate
biomarkers of the urogenital system. J Proteom Res 10: 941-953.
44.
Schiza CG, Jarvi K, Diamandis EP, Drabovich AP
(2014) An emerging role of TEX101 protein as a male infertility biomarker.
EJIFCC 25: 9-26.
45.
Fujihara Y, Tokuhiro K, Muro Y, Kondoh G, Araki Y,
et al. (2013) Expression of TEX101, regulated by ACE, is essential for the
production of fertile mouse spermatozoa. PNA Sci U S A 110: 8111-8116.
46.
Dimitrios K, Schiza C, Brinc D, Soosaipillai A,
Theano DK, et al. (2017) Preclinical evaluation of a TEX101 protein ELISA test
for the differential diagnosis of male infertility. BMC Med 15: 60.
47.
Freour T, Com E, Barriere P, Bouchot O, Jean M, et
al. (2013) Comparative proteomic analysis coupled with conventional protein
assay as a strategy to identify predictors of successful testicular sperm extraction
in patients with non-obstructive azoospermia Andrology 1: 414-420.
48.
Bieniek JM, Drabovich AP, Lo KC (2016) Seminal
biomarkers for the evaluation of male infertility. Asian J Androl 18: 426-433.
QUICK LINKS
- SUBMIT MANUSCRIPT
- RECOMMEND THE JOURNAL
-
SUBSCRIBE FOR ALERTS
RELATED JOURNALS
- Advance Research on Endocrinology and Metabolism (ISSN: 2689-8209)
- Journal of Cancer Science and Treatment (ISSN:2641-7472)
- Journal of Blood Transfusions and Diseases (ISSN:2641-4023)
- International Journal of Diabetes (ISSN: 2644-3031)
- Archive of Obstetrics Gynecology and Reproductive Medicine (ISSN:2640-2297)
- Journal of Psychiatry and Psychology Research (ISSN:2640-6136)
- International Journal of Medical and Clinical Imaging (ISSN:2573-1084)